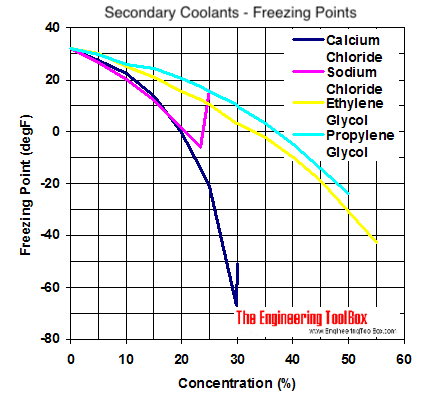
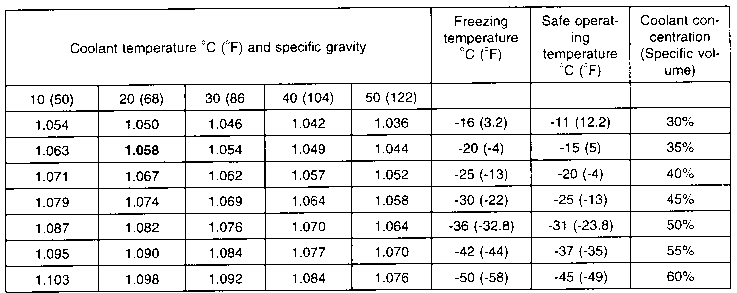
Ethylene glycol is used as an antifreeze agent. Calculate the amount of ethylene glycol to be added to 4 kg of water to prevent it from freezing at - 6^∘ C.(Kf for

Antifreeze Refractometer Displaying in Fahrenheit for Checking Freezing Point of Automobile Antifreeze Systems and Battery Fluid Condition. Battery Acid, Glycol, Coolant, Antifreeze Tester: Amazon.com: Industrial & Scientific