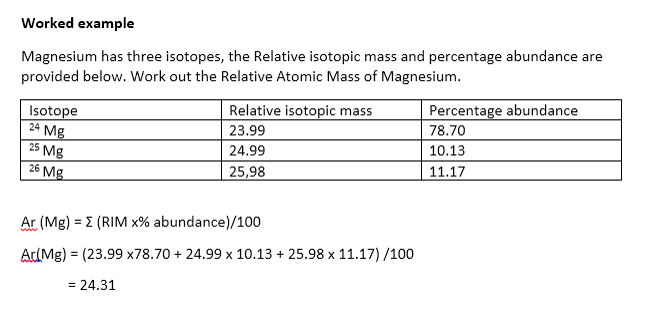
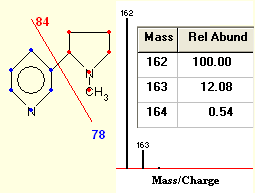
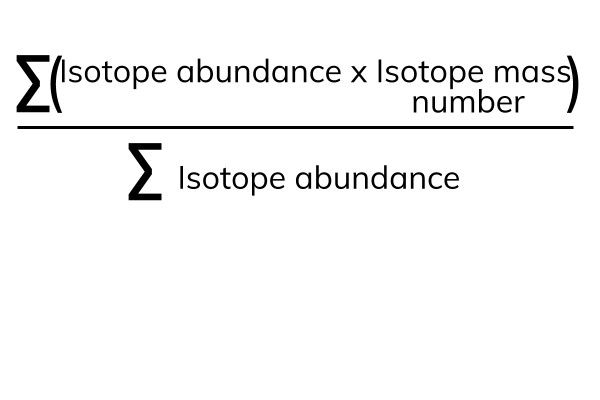
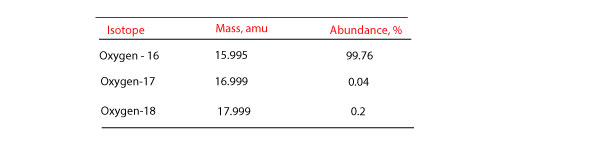
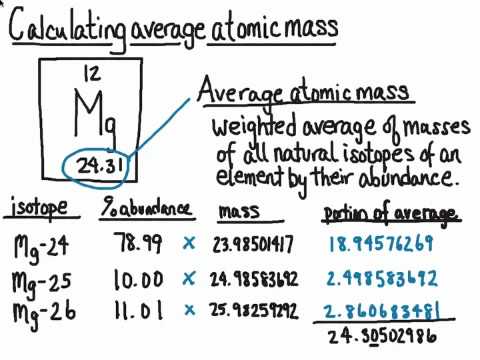
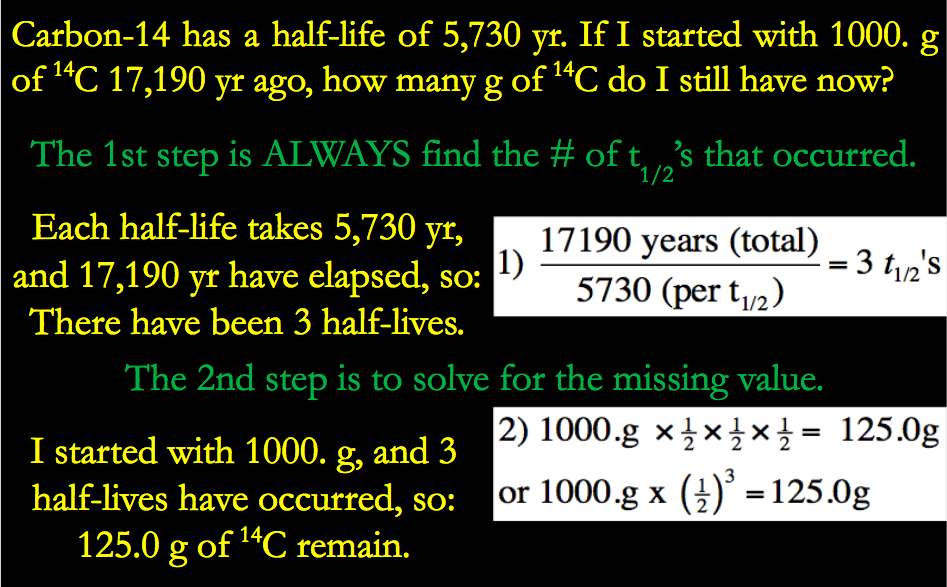What is an Isotope? | Examples, Types & How to Identify an Isotope - Video & Lesson Transcript | Study.com
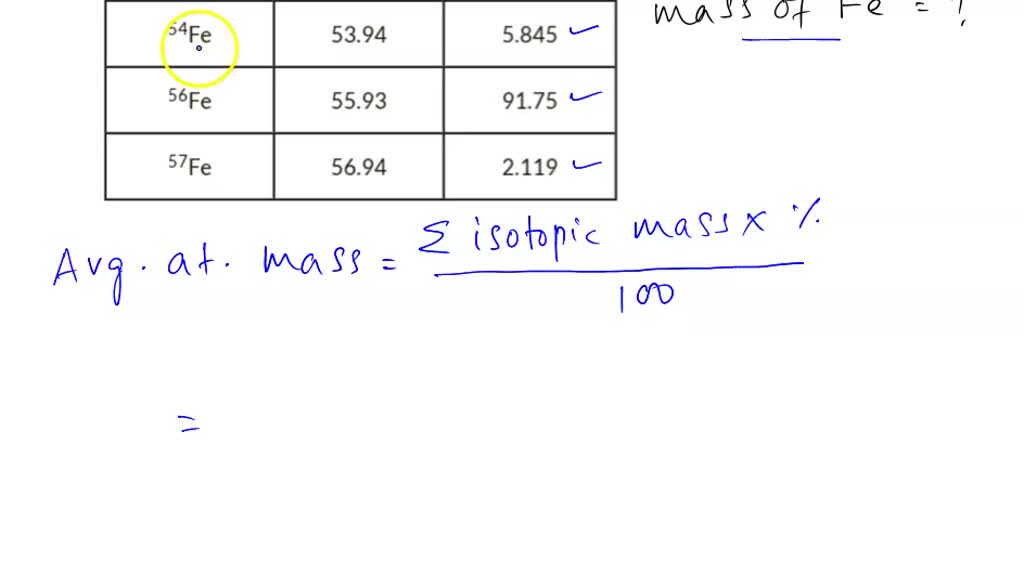
SOLVED: Calculate the average atomic mass of iron using the isotopes in Table 2 below: Table 2. Stable Isotopes of Iron Isotope Mass (amu) % Abundance 54Fe 53.94 5.845 56Fe 55.93 91.75 57Fe 56.94 2.119