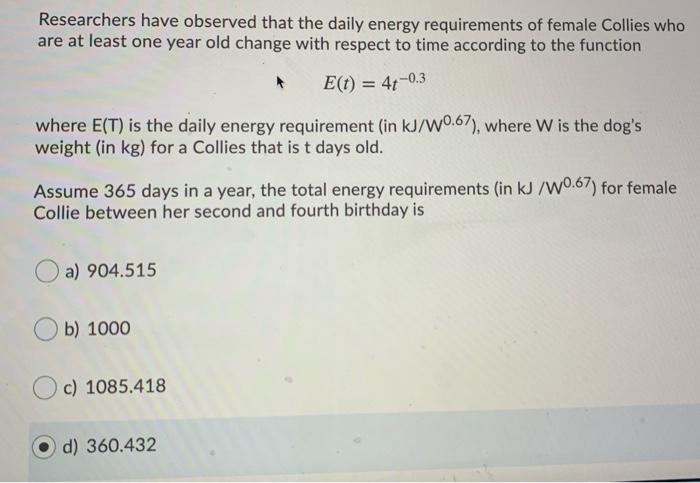
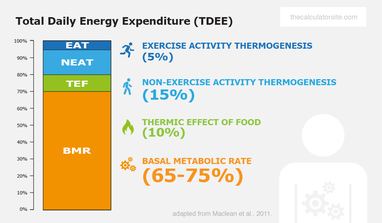
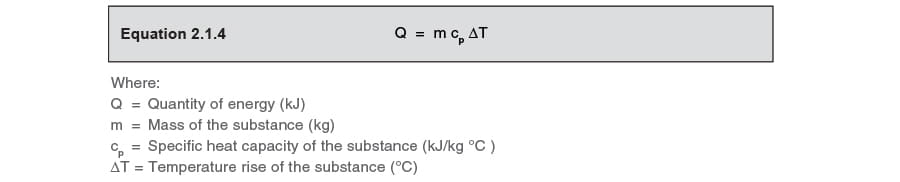
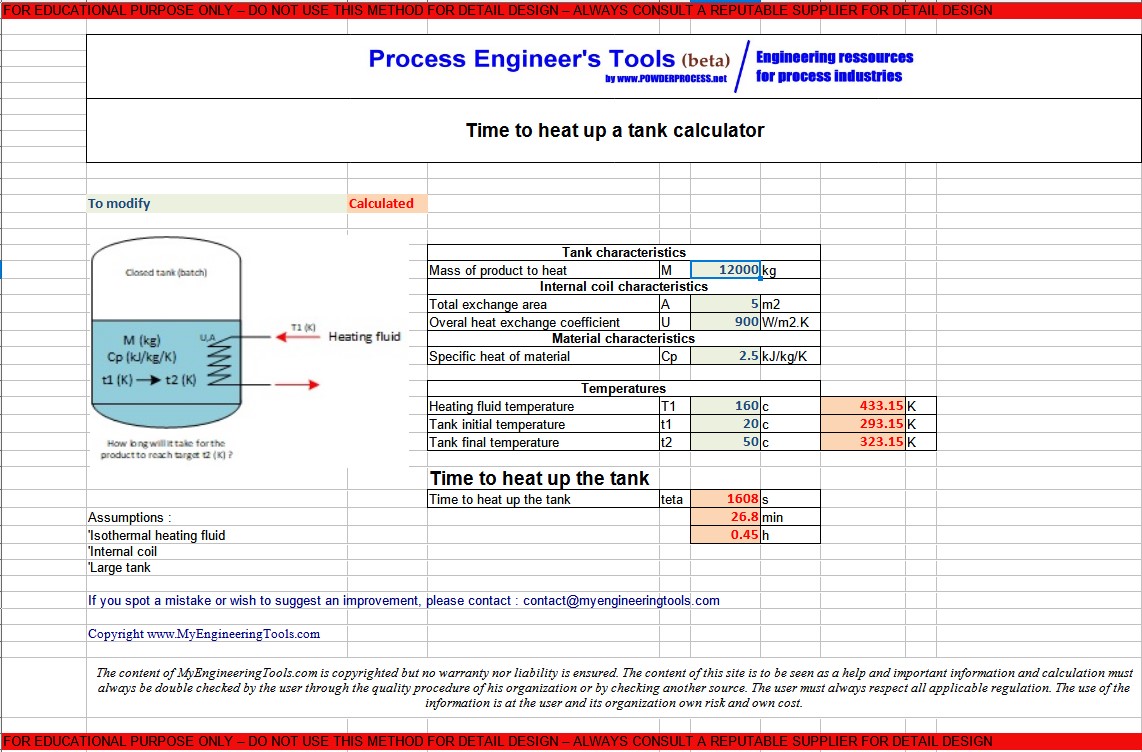
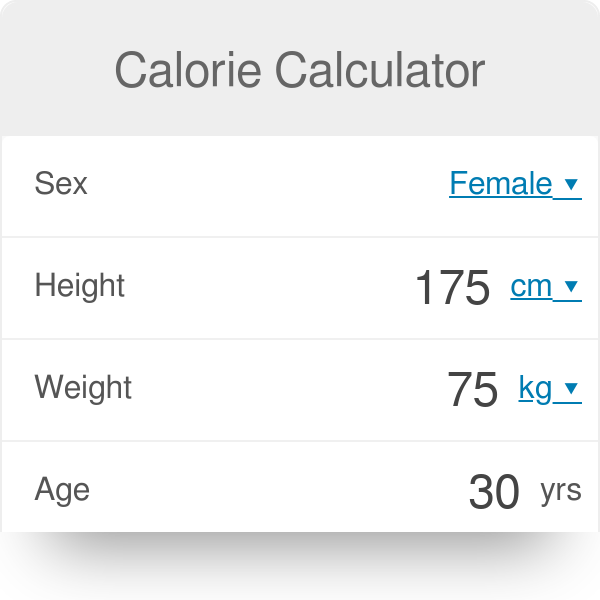
SOLVED: Calculate the amount of heat, in Kilojoules, needed to convert 75.0g of ice starting at -20.0C and ending as steam at 120.0C. specific heat of liquid water = 4.184 J/gC specific

Estimating Energy, Protein and Fluid Requirements For Adult Clinical Conditions | PDF | Body Mass Index | Dietitian