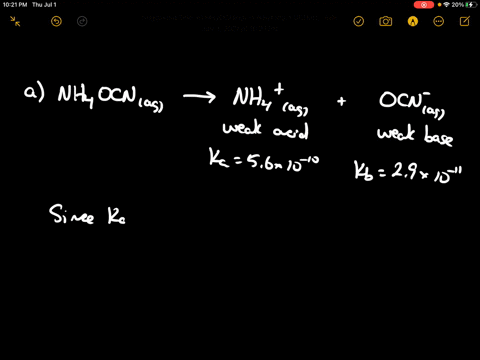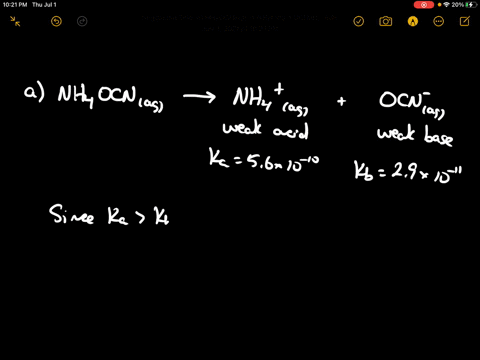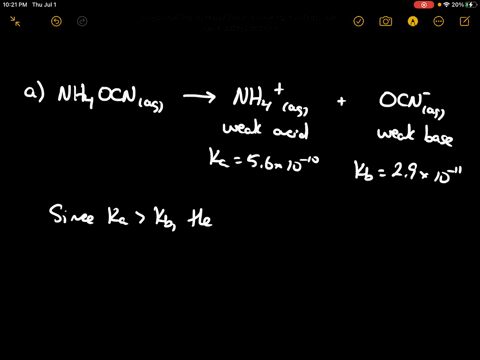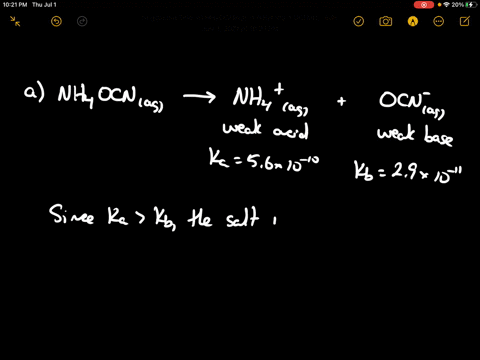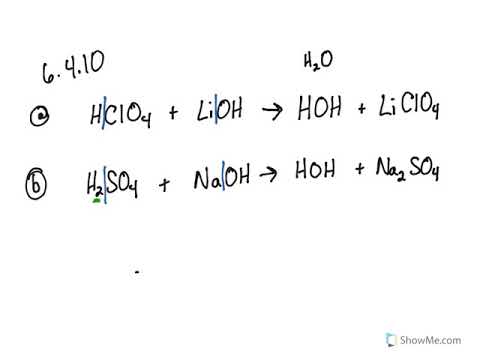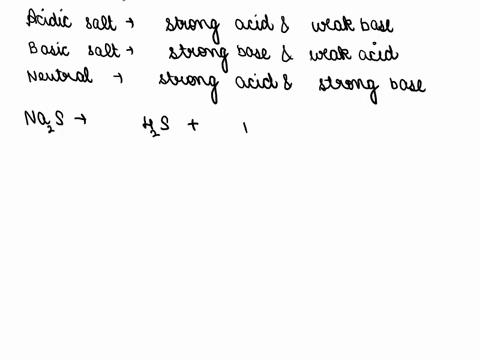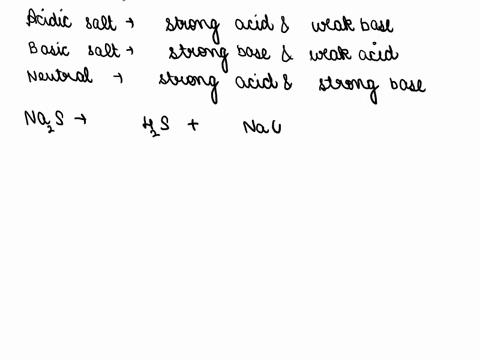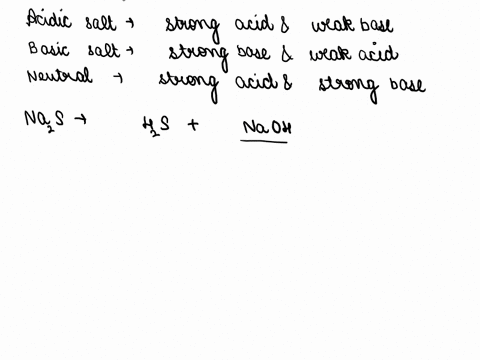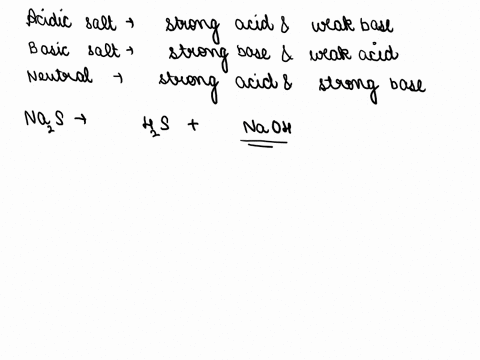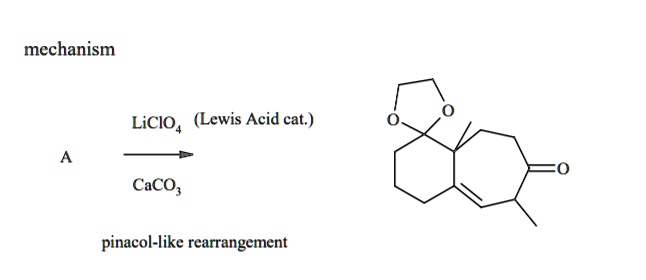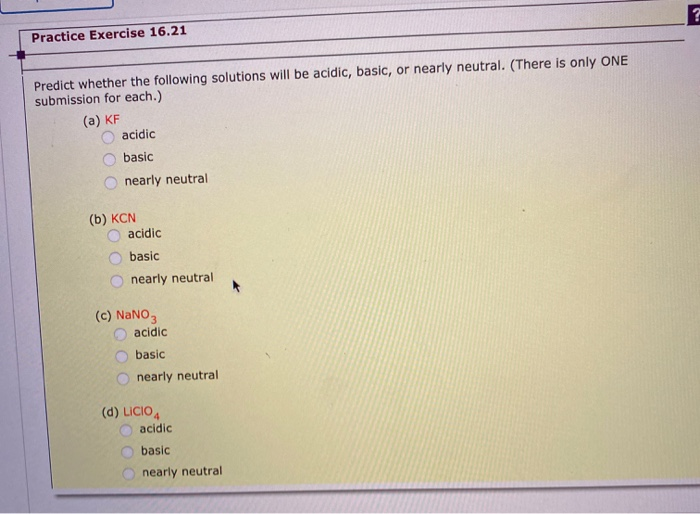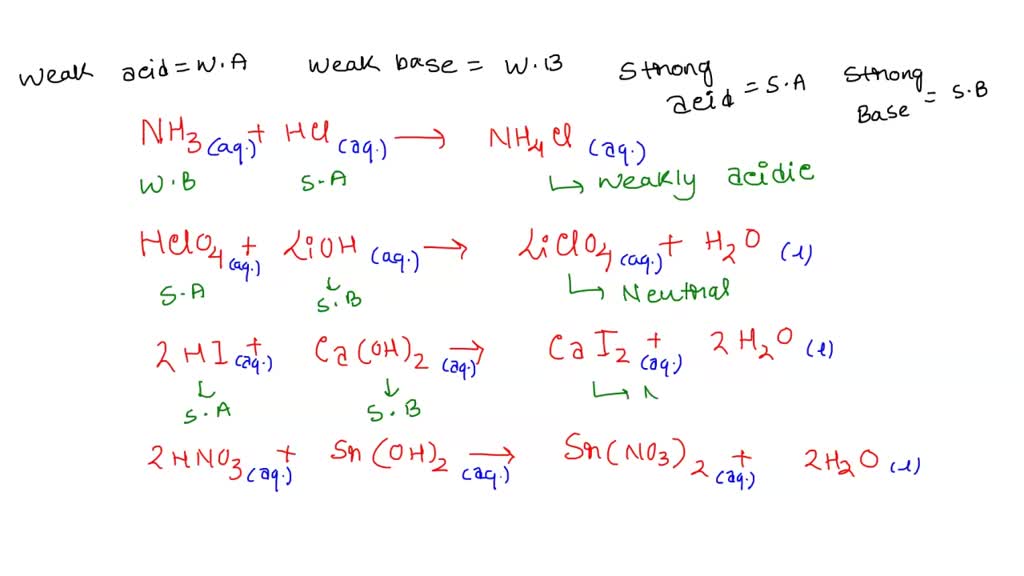
SOLVED: Of the following, which forms a weakly acidic solution? Assume all acids and bases are combined in stoichiometrically equivalent amounts. Select the correct answer below: NH3(aq)+HCl(aq)⇌NH4Cl(aq) HClO4(aq)+LiOH(aq)⇌LiClO4(aq)+H2O(l) 2HI(aq)+ ...
Spring 2009 CH302 Worksheet 5 Answer Key—How to Systematically Work Harder and Harder Acid Base Calculations Exactly the Same

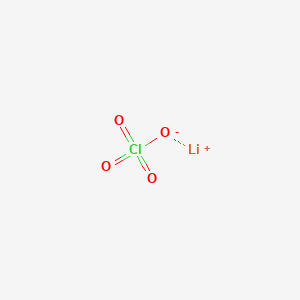
Solvate Structures and Computational/Spectroscopic Characterization of LiClO4 Electrolytes | The Journal of Physical Chemistry C
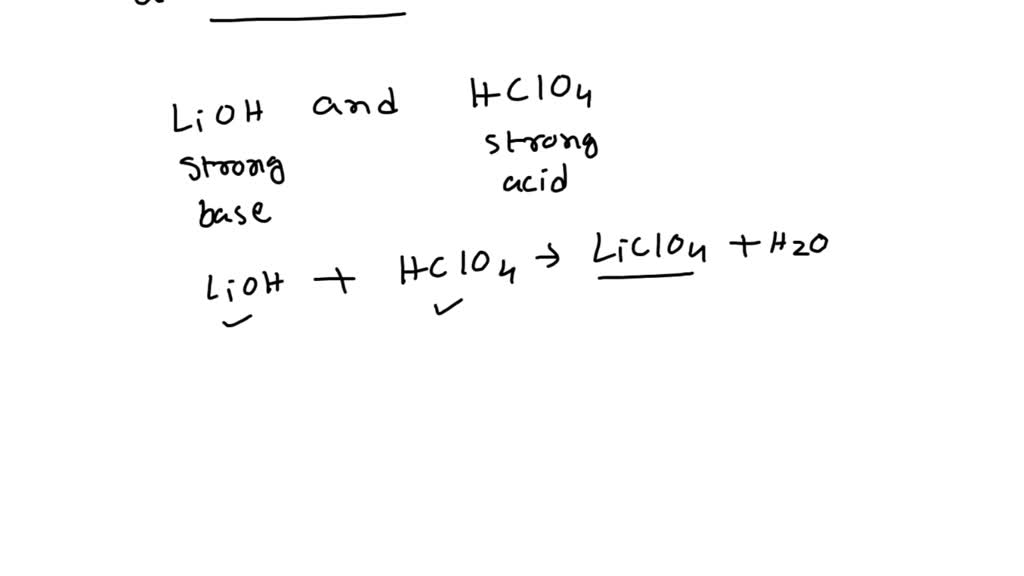
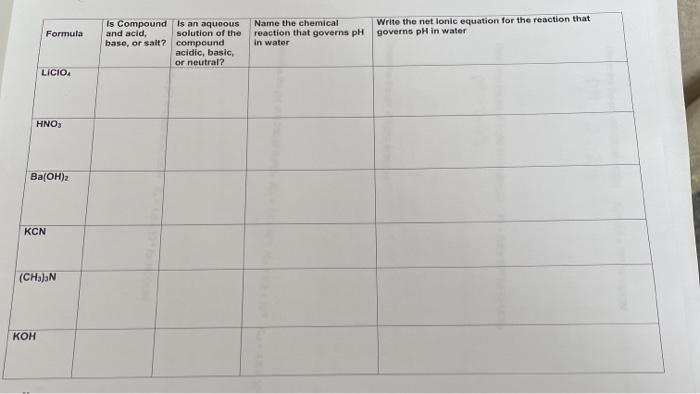
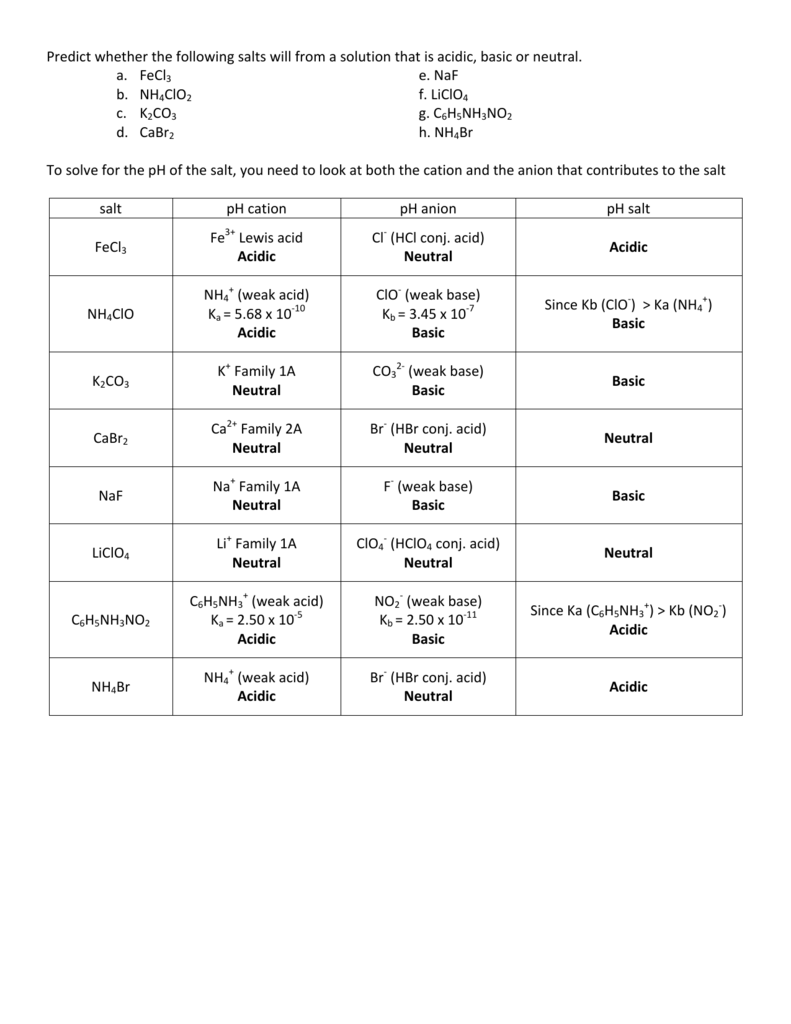
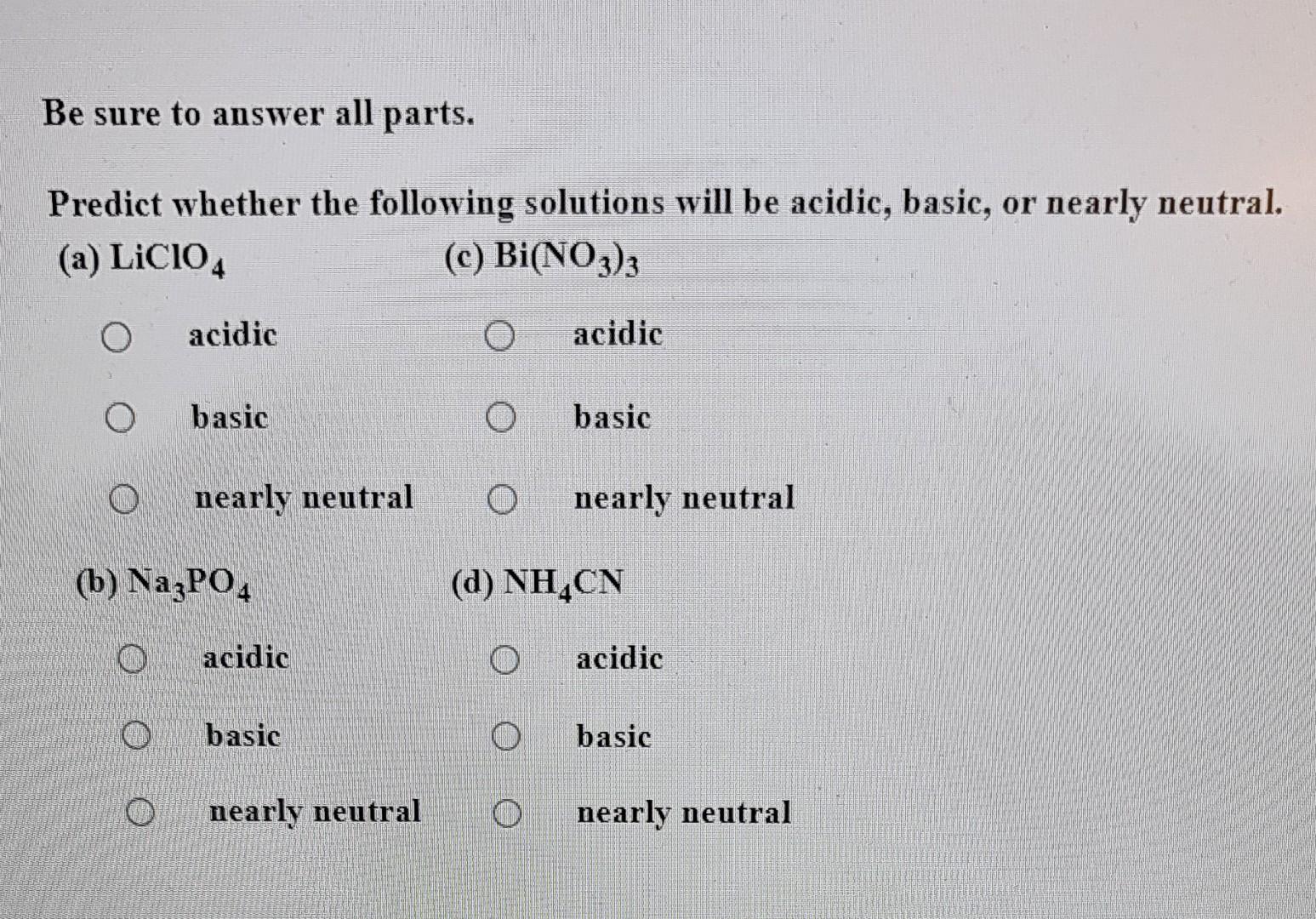
SOLVED: Predict whether the following solutions will be acidic; basic. Or nearly neutral (a) LiClO4 (c) Bi(NO3)3
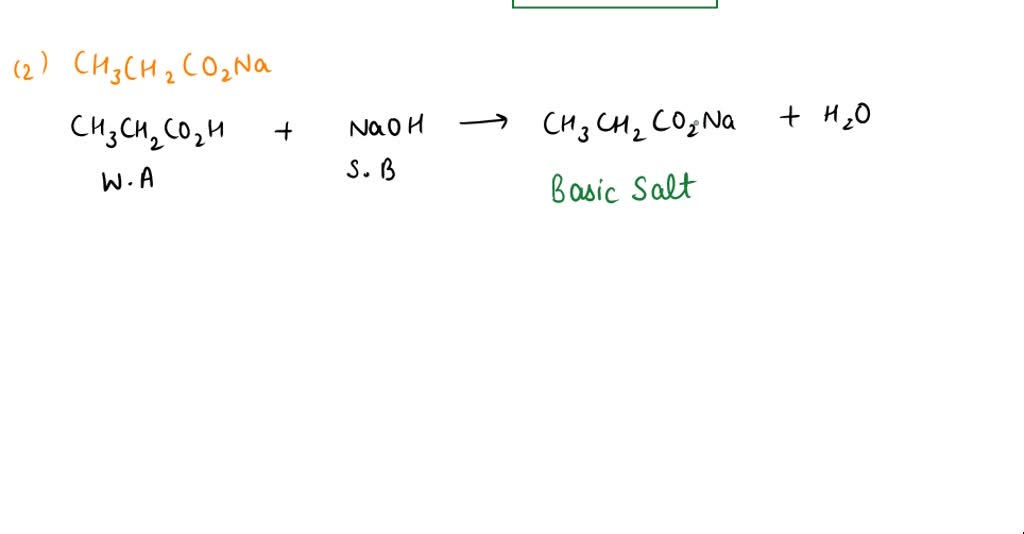
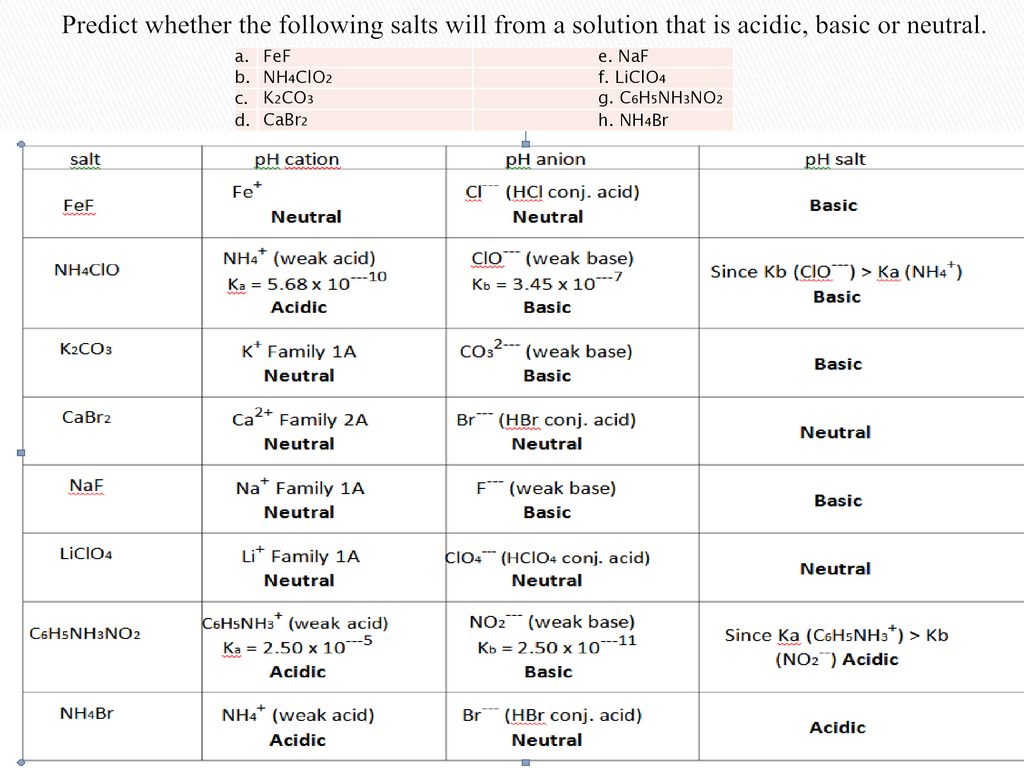
SOLVED: CLASSIFY THESE SALTS AS ACIDIC, BASIC, OR NEUTRAL. CH3CH2NH3Cl CH3CH2CO2Na HCO2K LiCLO4 CH3NH3NO3