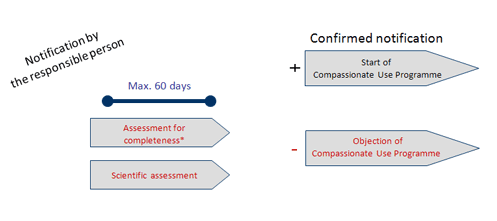
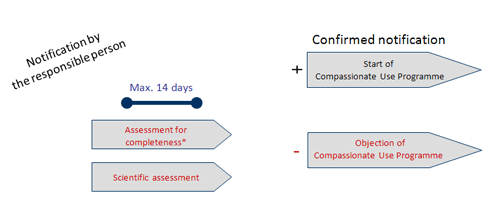
Compassionate Use Navigator Information for Physicians Application Process Overview Apply to a Drug Manufacturer

I'm Willing To Try Anything': Compassionate Use Access To Experimental Drugs And The Misguided Mission Of Right-To-Try Laws | Health Affairs

Compassionate use of drugs and medical devices in the United States, the European Union and Japan - ScienceDirect

Compassionate use of drugs and medical devices in the United States, the European Union and Japan - ScienceDirect

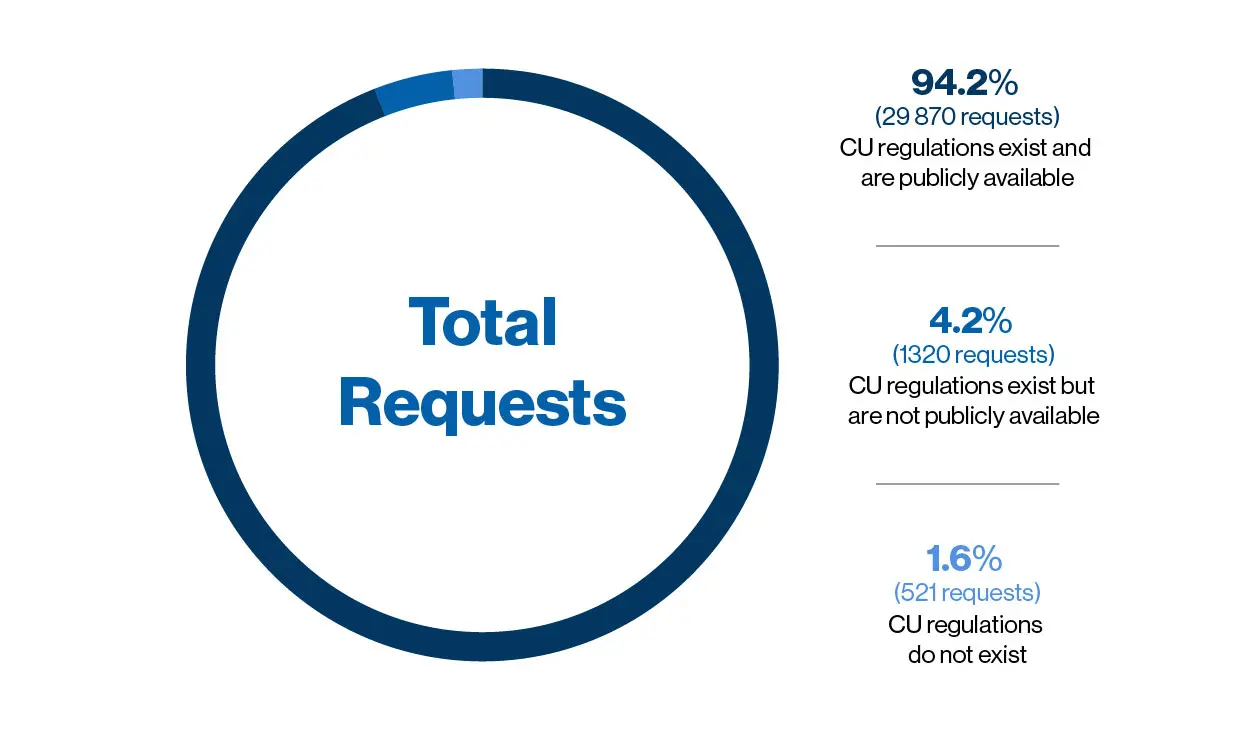
Manufacturer's Compassionate Use Policies: Companies with Posted Policies More Than Doubled Since September 2016

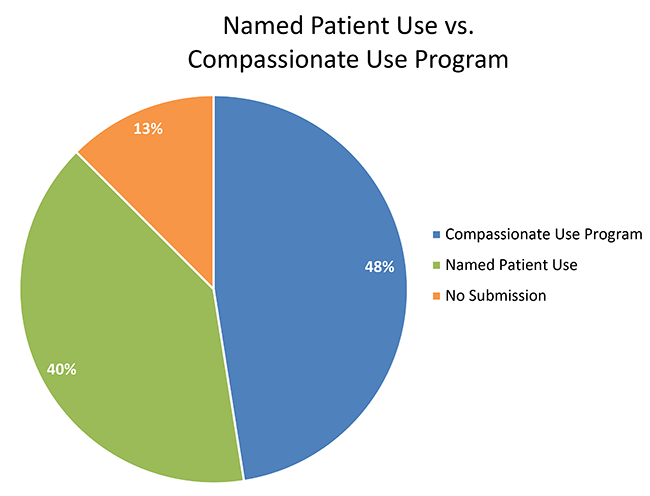
Understanding the challenges and ethical aspects of compassionate use of drugs in emergency situations Goyal PK, Mathur R, Medhi B - Indian J Pharmacol