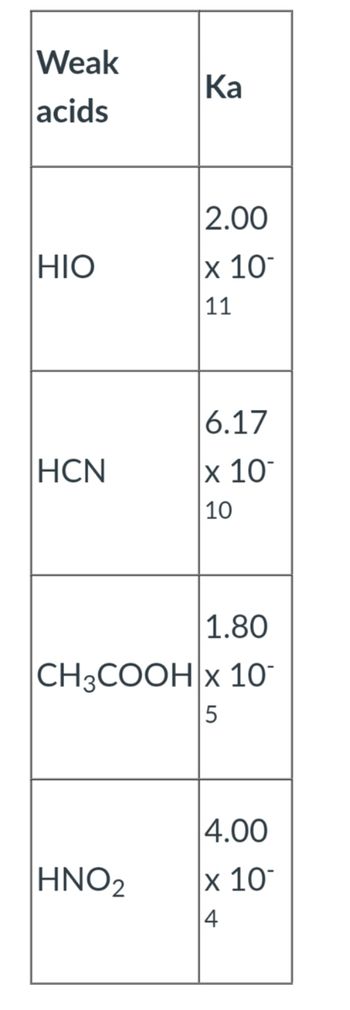
![SOLVED: Salts of hypoiodite ion behave as a weak base, undergoing hydrolysis in water according to the equation: IO- HzO = HIO + OH- Kb for hypoiodite ion 4.37x10-4 Calculate the [H+] SOLVED: Salts of hypoiodite ion behave as a weak base, undergoing hydrolysis in water according to the equation: IO- HzO = HIO + OH- Kb for hypoiodite ion 4.37x10-4 Calculate the [H+]](https://cdn.numerade.com/ask_images/9d80eb482c1a4947a4ffb060669f0832.jpg)
SOLVED: Salts of hypoiodite ion behave as a weak base, undergoing hydrolysis in water according to the equation: IO- HzO = HIO + OH- Kb for hypoiodite ion 4.37x10-4 Calculate the [H+]

Hion Calculator,12-Digit Large Display Office Desk Calcultors with Erasable Writing Table,Rechargeable Hand held Multi-Function Mute Pocket Desktop Calculator for Basic Financial Home School,White

SOLVED: (5) Calculate the open cell potential, Ecell, if the half-reaction CuzO(s) + HzO(l) + 2 e 2 Cu(s) + 2 OH-(aq) is coupled to a Ag/AgCl reference electrode. Standard conditions apply

Calculate the concentration of all the species present in a 0.100 M solution of hypoiodous acid. (Ka = 2.3 x 10-11) | Homework.Study.com

![Pangya] Hole In One Calculator Test - YouTube Pangya] Hole In One Calculator Test - YouTube](https://i.ytimg.com/vi/lrSLGxlc5KM/maxresdefault.jpg)